“Treat All” provides new hope as well as new challenges in the fight against HIV
 November 30, 2015
November 30, 2015 World Aids Day 2015 once again directs the public’s attention toward the ongoing HIV pandemic and the efforts to contain it. As the year draws to an end, we can see great progress over the past twelve months and a clear agenda for 2016.
This past May, we learned that the START study, sponsored by UNAIDS, was being prematurely concluded by study organizers because results were already so clear: Data showed that those who received anti-retroviral therapy (ART) immediately upon HIV diagnosis were 53% less likely to die during the trial or develop AIDS or a serious illness than those who waited for initiation of treatment.
The START study addressed a point that had been widely debated by the HIV healthcare community for years. That debate was swiftly resolved when Michel Sidibé, Executive Director of UNAIDS, concluded that, “Every person living with HIV should have immediate access to life-saving antiretroviral therapy. Delaying access to HIV treatment under any pretext is denying the right to health.”
It didn’t take long for the World Health Organization (WHO) to act on the findings. In September of this year, the WHO updated its treatment guidelines to include a “treat all” provision. It recommends that everyone with HIV is provided ART immediately after diagnosis. This is one of the most significant developments in the treatment of HIV over the last decade. Michel Sidibé explained that the policy change could help prevent 21 million AIDS-related deaths and 28 million new infections by 2030.
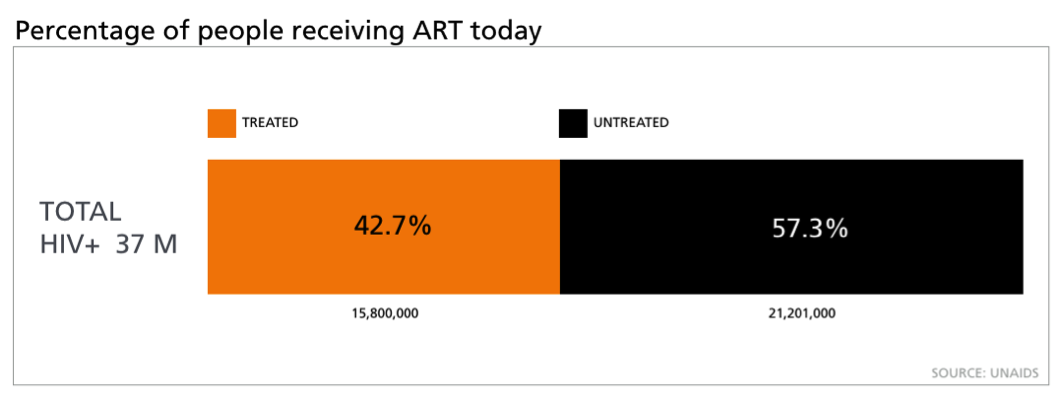
Of course, with the new WHO guidelines come new challenges for healthcare providers around the world. According to UNAID’s estimates, 57% of HIV positive adults do not receive treatment today. Based on that figure, the new WHO guidelines will mean proving ART to an additional 21 million people globally.
One of the greatest challenges in making “treat all” a reality is that the overwhelming majority of the untreated live in resource-limited settings. In developing nations around the world, healthcare organizations are working to find ways to increase access to ART, as well as to the viral load monitoring required for proper administration of the life-saving drugs. I’m happy to report progress on that front as well.
In September of this year, Cavidi was selected by the European Investment Bank (EIB) as a recipient of their Infectious Diseases Innovation Loan for 2015. The focus of the loan is our Ziva™ viral load monitoring system. Viral load monitoring is an essential part of HIV treatment, but, until now, it has been difficult to administer in near-patient settings. Ziva™ changes that by providing a medium throughput, fully-automated viral load monitoring system purpose-built to meet the requirements of decentralized labs like those found in district hospitals across the developing world. Its unique Reverse Transcriptase (RT) technology makes fully-automated gold-standard results possible for decentralized testing.
The recognition of the EIB is a great honor for Cavidi, but an even bigger win for people infected with HIV around the world. While the €10 million loan is only part of the investment required to get Ziva™ to market, it will go far to provide access to viral load monitoring sooner for millions living with HIV.
All things considered, 2015 has been a pivotal year in the battle against HIV and cause for renewed optimism. The new WHO guidelines mark a significant shift in the global treatment mindset. The HIV treatment community has overcome great challenges to reach this point where “treat all” is the official policy moving forward. The financial and logistic challenges we now face to implement the policy are significant, yet dwarfed in comparison. Addressing these new challenges will be our focus at Cavidi for 2016 and beyond.


Reader Comments